Regulatory & Safety
Our regulatory operations solutions reduce complexity, improve efficiency and enhance compliance. Streamline processes, reduce costs, and foster collaboration through our proven methodologies. Make data-driven decisions and accelerate time-to-market with an integrated regulatory system. Seamlessly migrate your data, eliminate redundancy, and overcome complexities. Ensure robust structured data and GxP processes to stay compliant and inspection ready.
By leveraging innovative solutions in the Pharmacovigilance domain, Pharmaceutical Companies can better contribute to the safety of patients and improve public health outcomes.
The Regulatory House of Excellence
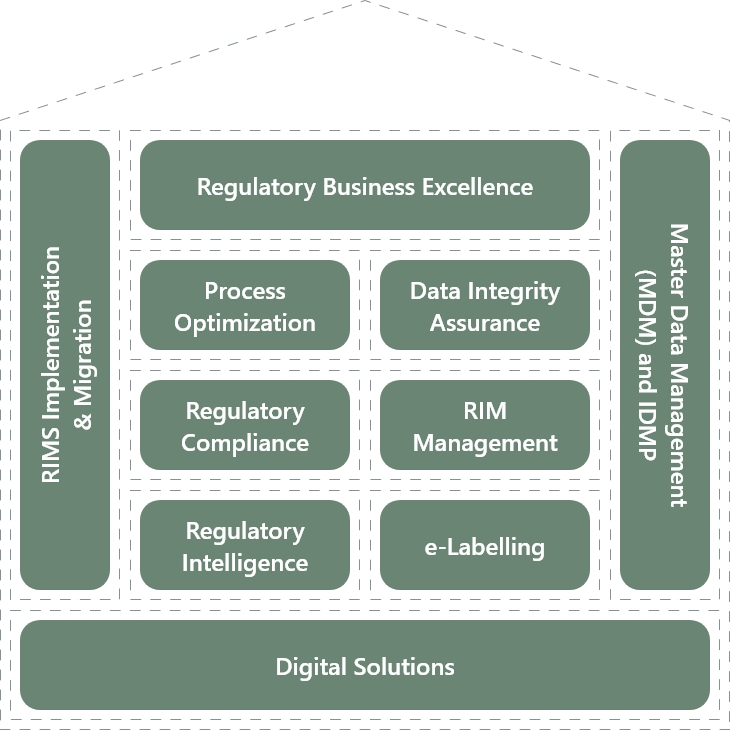
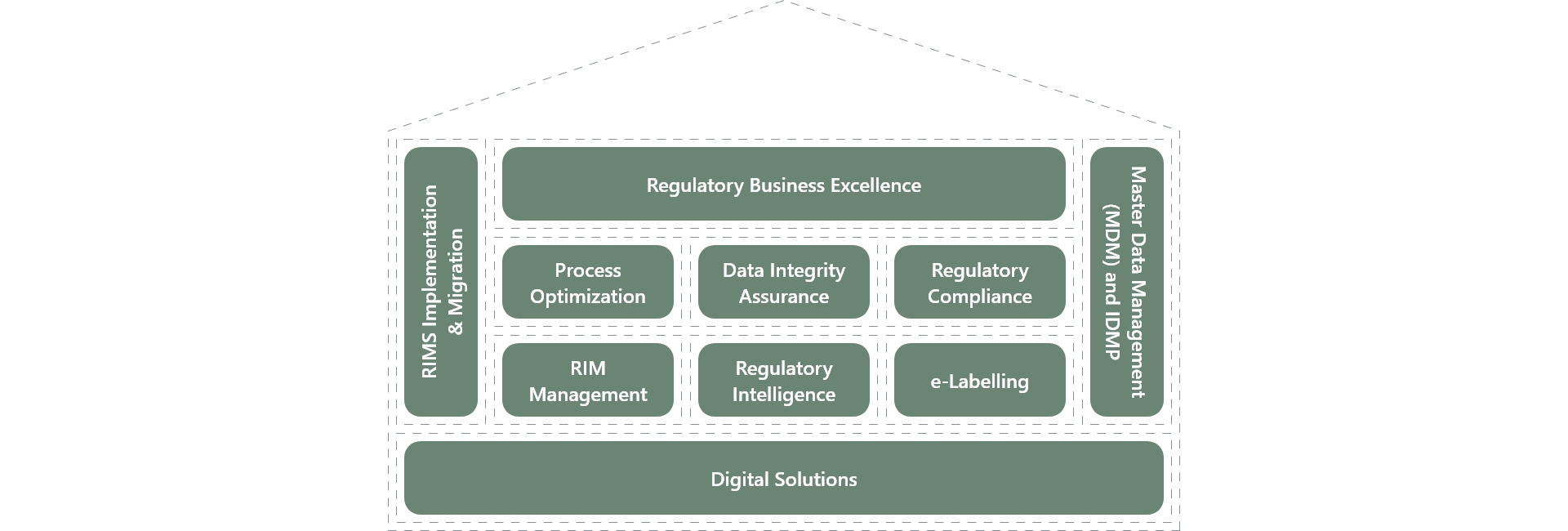
Offerings
Process Optimization
We offer a comprehensive tool set to increase the speed and effectiveness of any process through Lean Six Sigma© methodologies.
Data Migration
We have performed over 70 data migrations within GxP environments. Our experience spans migrating data to registrations, submissions, submissions archive, IDMP, and publishing modules.
Data Integrity Assurance
We can help you collect and organize data from separate systems across a company, including unstructured data.
Regulatory Compliance
We can evaluate Data, Processes and Systems against industry standards and best practices and propose a tailored roadmap to thrive operational excellence and compliance.
RIM Management
Through our Veeva Vault Admin© certified consultants, we have extensive expertise in Veeva RIM Management to support the administration of Veeva RIM and training needs.
Regulatory Intelligence
Through dedicated AI-powered solutions, BASE can help customers screening through Reg Intel sources and performing tailored impact assessments.
e-Labelling
Our experienced consultants help clients prepare for PLM implementation and provide full support in their journey towards full electronic product information.
Publishing
We offer expert support with the implementation and optimization of client’s Publishing tools and provide application services and management as needed.
Master Data Management (MDM)
Through our proven methodology, our experienced BASE consultants can support the definition and implementation of structured MDM models within our customer’s organization.
xEVMPD
We offer expert support with the implementation and optimization of Veeva xEVMPD and provide application services and management consulting for the end-to-end Veeva journey.
SPOR/DADI
We have hands-on expertise in data cleansing, enrichment and preparation for SPOR/DADI implementation.
IDMP Journey
We can run an assessment to evaluate the IDMP preparedness and provide tools tailored to customer’s needs to ensure IDMP compliance.
Safety Data Management 
We streamline the process of your data, reducing efforts and expediting the decision making process.
Safety Compliance
To keep your Pharmacovigilance System compliant, it is crucial to implement solutions to facilitate metrics calculation and maintain a robust oversight at global and local level.
Safety Process Optimization
Intelligent automation can optimize and harmonise pharmacovigilance processes, providing a more efficient way to analyse safety information and making a positive impact on the quality of safety analyses.
Veeva Value Journey
We streamline Regulatory and Safety processes, ensure compliance, enhance operations, and uphold the highest safety and quality standards. Learn more about the Veeva Value Journey.
Digital Solutions
We’ve enhanced efficiency with Automated Document Review, innovated with IDMP Metadata Extraction for precise data retrieval, and implemented the HA Q&A Bot for instant regulatory inquiries. Learn more about Digital Solutions.

